 “Soft Matter” refers to those materials constituted by structural units bigger than atoms, that typically display a collective behavior. Colloids, emulsions, pastes, gels, liquid crystals and surfactants are some examples of soft materials. They all have in common two characteristics: non-linear responses when a stress is applied; slow and non-equilibrium response. In our laboratories, we are investigating colloidal suspensions behavior, dense emulsions, biological samples and liquid crystals.
“Soft Matter” refers to those materials constituted by structural units bigger than atoms, that typically display a collective behavior. Colloids, emulsions, pastes, gels, liquid crystals and surfactants are some examples of soft materials. They all have in common two characteristics: non-linear responses when a stress is applied; slow and non-equilibrium response. In our laboratories, we are investigating colloidal suspensions behavior, dense emulsions, biological samples and liquid crystals.
Yield-stress fluids
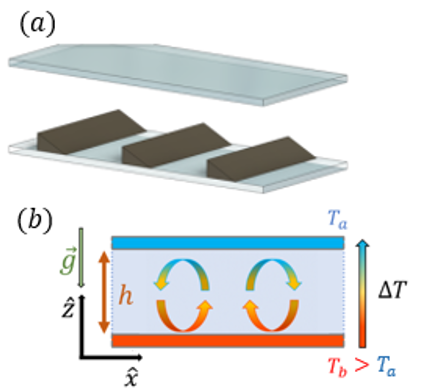
Yield-stress fluids are soft materials whose behavior depends on the stress applied with a threshold mechanism: below the threshold (yield point) they behave like an elastic solid; above the threshold …
